Measured out 1ml of water in a 10ml measuring cylinder and placed into the test tube labelled 2.
Marble chips and hydrochloric acid experiment surface area method.
2 to investigate the effect of temperature of sodium thiosulphate in a reaction with hydrochloric acid.
To observe the effect of surface area particle size on the rate of reaction between marble chips calcium carbonate and hcl.
Calcium chloride solution is also formed.
Measured 5ml of hydrochloric acid in the 10ml measuring cylinders and placed into each beaker separately.
To investigate the effect of concentration of sodium thiosulphate in a reaction with hydrochloric acid.
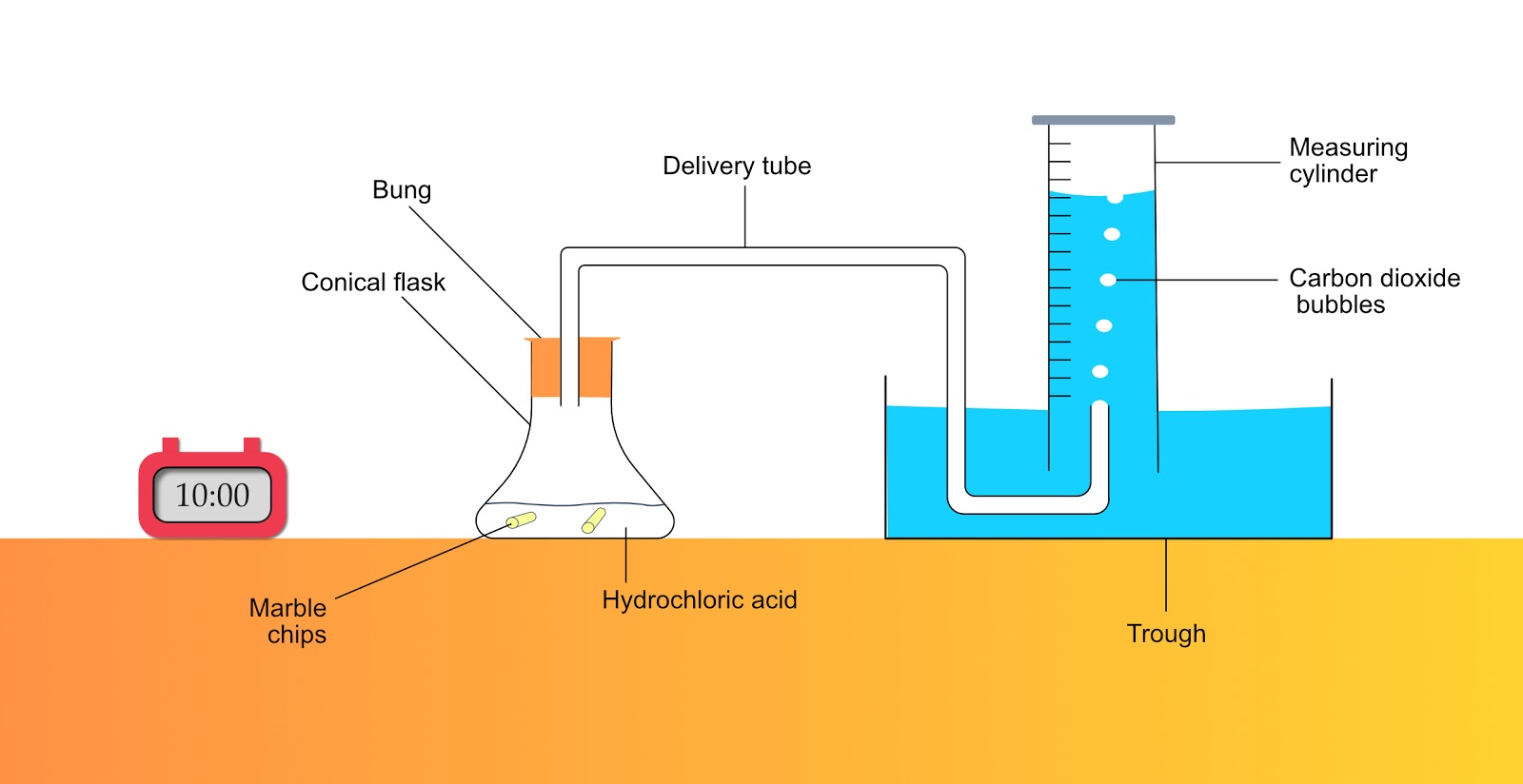
Investigating the rate of reaction between marble chips calcium carbonate and hydrochloric acid aim.
An investigation of the reaction between marble chips and hydrochloric acid.
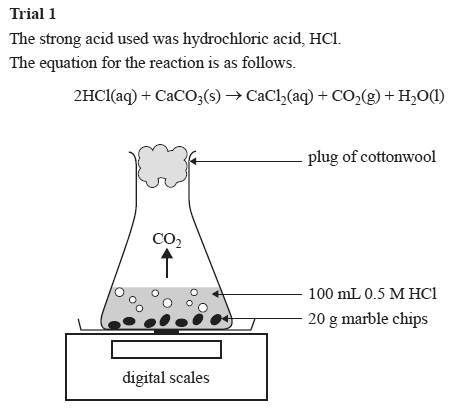
Hcl calcium carbonate calcium chloride carbon dioxide water.
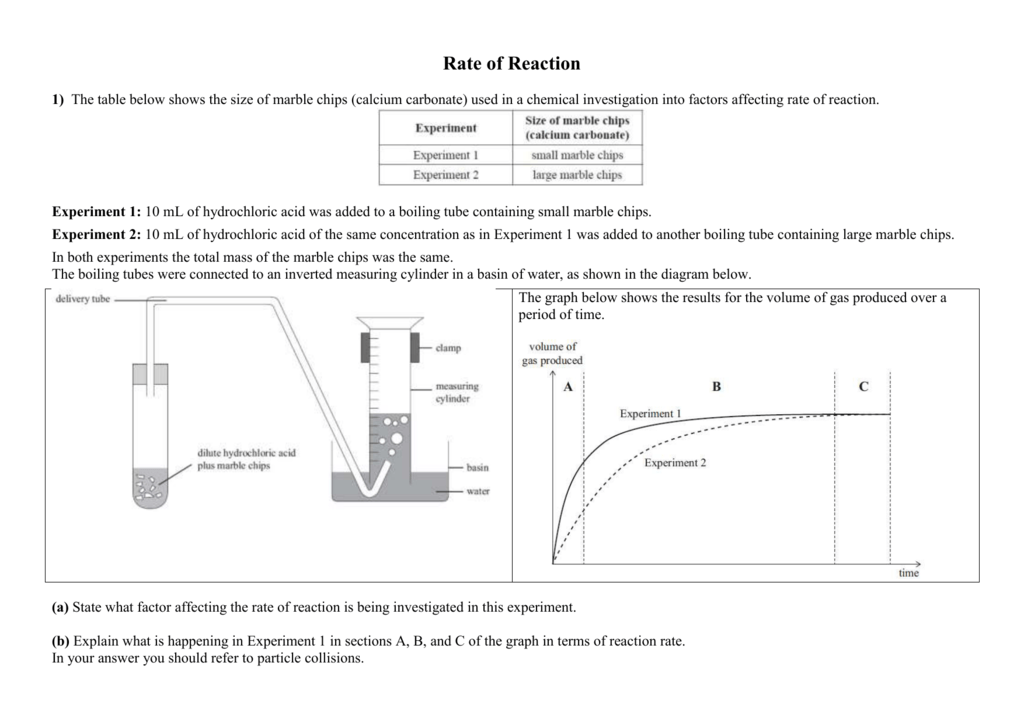
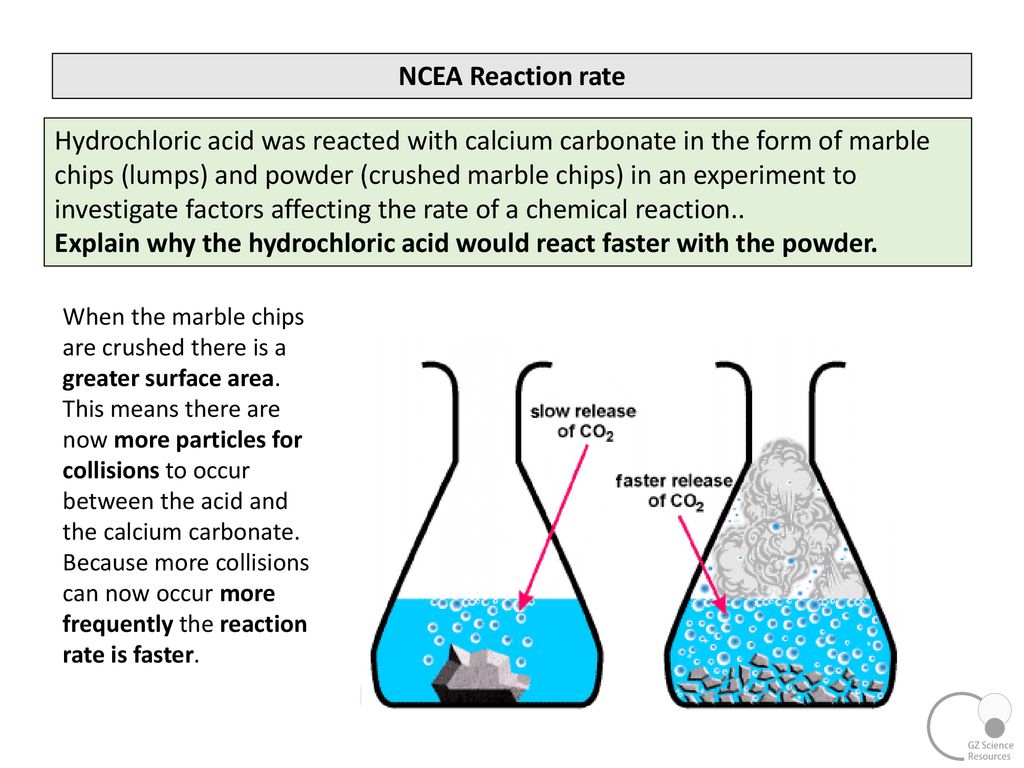
In the investigation i am going to find out how the surface area affects the rate of reaction by measuring the amount of gas produced and weight loss in a reaction between small large pieces of marble chips calcium carbonate and hydrochloric acid per minute.
Marble chips placed onto pieces of paper.
Marble chips react with dilute hydrochloric acid to produce carbon dioxide gas.
As the marble chips react with.
And dilute hydrochloric acid calcium carbonate may be used in the form of marble chips.
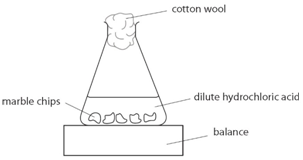
I will weigh out one gram of marble chips using a balance and put it in a conical flask and add to it a concentration of 50cm3 using water and hydrochloric acid.
Plugged in scientific scales and weighed out 1g of marble chips for each test tube.
Contains the marble chips hydrochloric acid and the water that will make the reaction.
Finding the rate of reaction of marble chips and hydrochloric acid changing the surface area.
Finding the rate of reaction of marble chips and hydrochloric acid changing the surface area.
Smaller chips have a larger surface area.
The surface area isn t always the same so even though the mass of the marble.
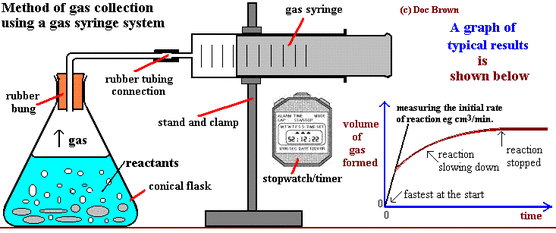
Using the apparatus shown the change in mass of carbon dioxide can be measure with time.